MONITORING &
CONSUMABLES
MUCH MORE THAN AN INTEGRAL
SOLUTION PROVIDER
Indicators for routine monitoring act as a critical benchmark for checking and recording RUMED performance. These benchmarks enable RUMED supervisors to have additional quality control to check that all medical devices and loads exceed the acceptable reference threshold, even taking into account the process conditions in close contact with the medical devices.
Monitoring for process validation is a set of Indicators that allow microbiological and physico-chemical validation of medical device and other device reprocessing processes according to the validation standards of each process. In this case, the processes will be validated following the requirements of the validation standard by addressing different approaches to comply with the limits required in each case.
PRODUCTS
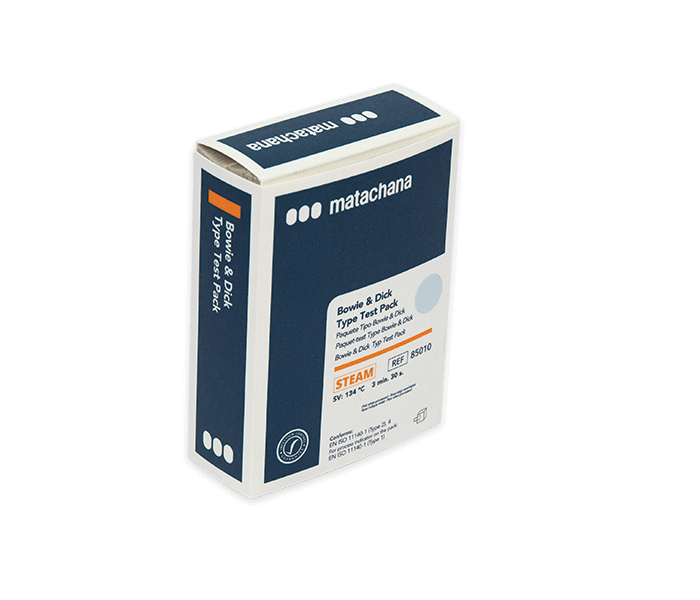










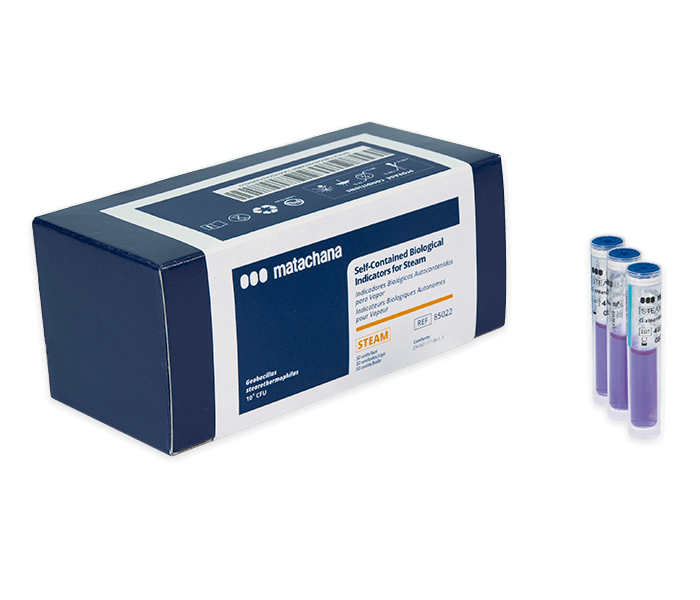


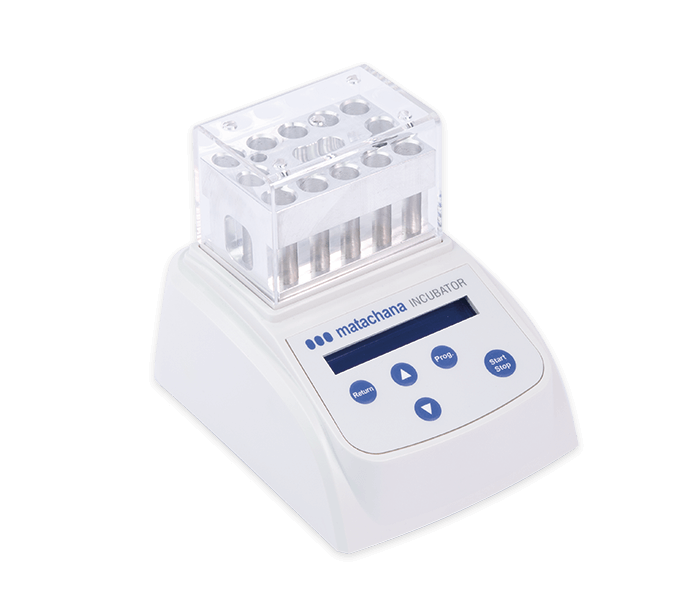

LABELLER (81373.5)





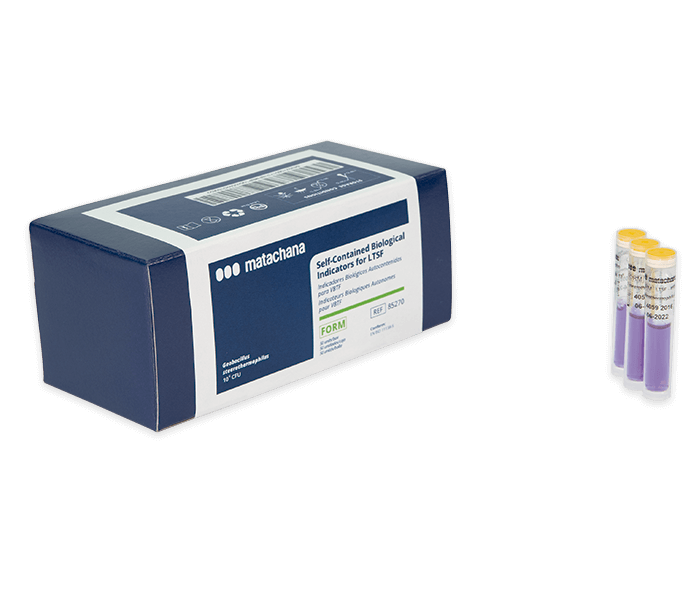


LABELLER (81373.5)






LABELLER (81373.5)